Modernizing sample operations with QuartzBio’s Sample Intelligence solution
About the Client
A Top 5 global pharmaceutical company managing more than 100 concurrent clinical trials came to QuartzBio seeking to increase their R&D pipeline capacity without having to add headcount. They needed to move faster, operate smarter, and scale rapidly, all while keeping compliance and data quality intact.
This case study follows their journey from fragmented, manual sample operations to a fully connected, AI-powered workflow. The study showed that automation and unified sample intelligence can deliver both immediate operational improvements and long‑term portfolio and team scalability across complex global clinical programs.
The Challenges
Managing biospecimens across dozens of global clinical programs posed operationally challenging, and the client’s sample operations teams were stretched thin. The tools and systems they were using were not keeping pace with their needs.
Three core pain points stood out:
- Fragmented systems: Paper logs, spreadsheets, and disconnected platforms created silos that made it difficult to get a clear, unified view of sample data.
- Time‑consuming manual workflows: Routine tasks like copy-pasting files, consolidating informed consent data, and reconciling records across systems consumed hours that could have been spent on higher-value work.
- Lack of sample lifecycle visibility: Teams had no reliable way to track sample counts, flag missing samples, project upcoming collections, or confirm consent completeness.
The path forward required two things: (1) unified, harmonized data that all stakeholders could access, and (2) a measurable reduction in the manual effort required to keep that data current.
The Solution: QuartzBio’s Sample Intelligence Solution
The client team implemented QuartzBio’s Sample Intelligence solution, the industry’s only connected, AI-powered solution spanning the entire clinical sample lifecycle—from collection to assay insight.
The solution combines deep domain expertise in data management and sample lifecycles with compliant (21 CFR Part 11, GxP / ICH E6 (R3), GDPR, HIPAA) business intelligence. The secure, interoperable platform provides automated data ingestion, harmonization, query tracking, sample lifecycle monitoring, consent tracking, and real‑time analytics powered by AI agents to unify, monitor, and maintain high‑quality biospecimen data across studies and vendors.
Throughout the case study period, the Sample Intelligence solution delivered broad impact across four client use cases:
Use Case 1: 360° Sample Visibility
- A harmonized Master Sample Inventory integrating lab, EDC, and consent data.
- Full visibility into shipped, analyzed, missing, or stable/unstable samples.
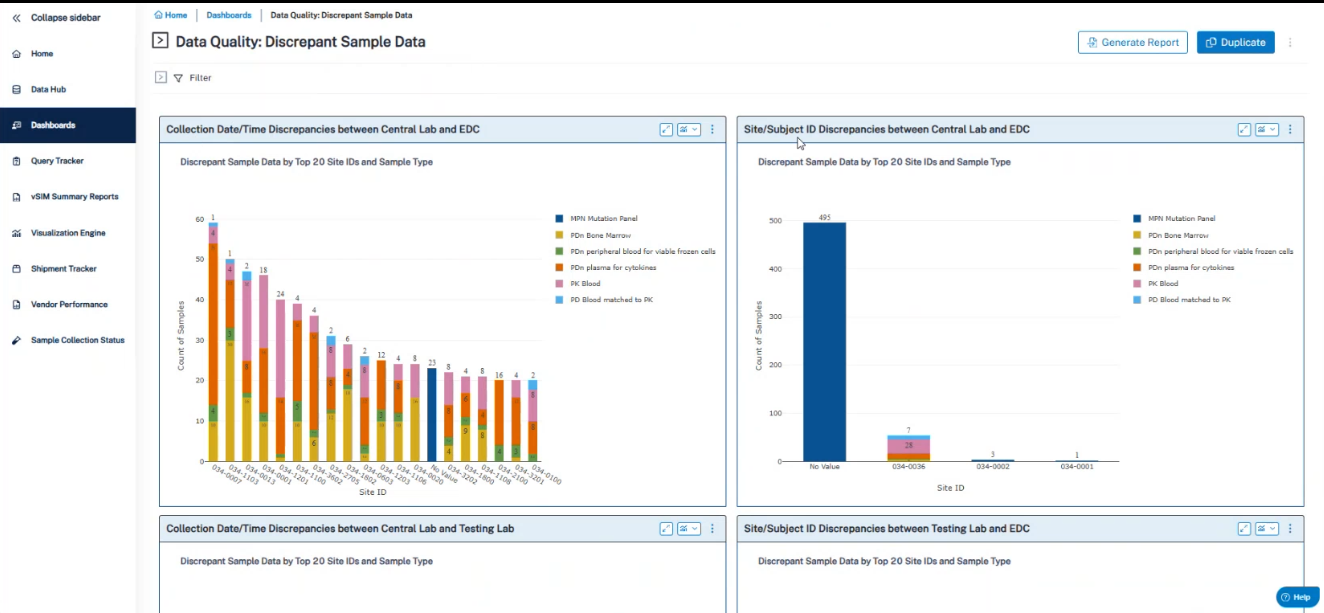
Use Case 2: Sample Data Quality
- Automated detection of discrepancies across files.
- Automated query tracking, resolution insights, and root‑cause visibility.
- Identification of previously unknown data issues, improving data integrity.
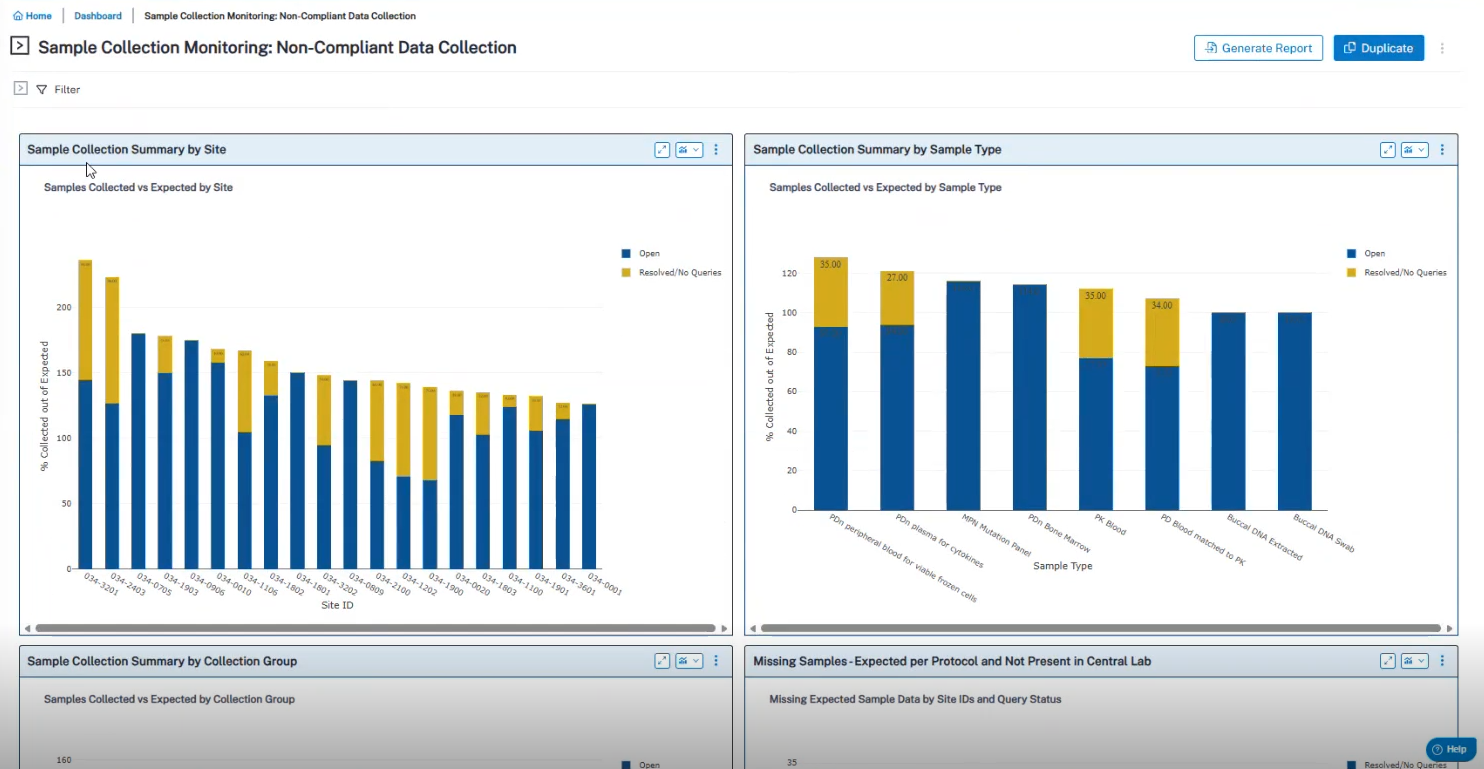
Use Case 3: Sample Collection Monitoring & Compliance
- Identification of samples not collected and visibility into expected vs. collected counts.
- Projections of upcoming sample collections.
- Support for ICH E6(R3) through timely detection of compliance risks.
Use Case 4: Sample Logistics & Testing Monitoring
- Real‑time tracking of sample status, location, and chain of custody.
- Monitoring of sample stability, duplicate testing, shipments, and testing status.
Outcomes
The teams achieved dramatic efficiency improvements, including:
- 85% reduction in time spent on sample‑related tasks (Table 1)
- 100% detection rate for missing samples and automated query generation.
- Significant reductions in data ingestion, reconciliation, consent tracking, and Excel®‑based processes.
| Task | Time Required Without QuartzBio | Time Required With QuartzBio | Impact |
|---|---|---|---|
| Data ingestion & file creation | 527 min | 24 min | 82% faster |
| Sample collection monitoring | 120 min | 2 min | 98% faster • 100% missing sample detection |
| Query tracking automation | 120 min | 10 min | 92% faster • 100% automated query generation |
| Manual Excel® manipulation | 143 min | 8 min | 87% faster |
| Consent tracking completeness | 40 min | 5 min | 88% faster • 100% consent documentation |
| Data reconciliation | 120 min | 3 min | 98% faster |
Capacity Expansion*
Because teams were spending dramatically less time on routine tasks, they could take on significantly more work — without adding headcount. Study capacity doubled across both early-phase (Phase I) and late-phase (Phase II/III) trials, representing an increase of 100+ trials managed by the same team (Figure 1).
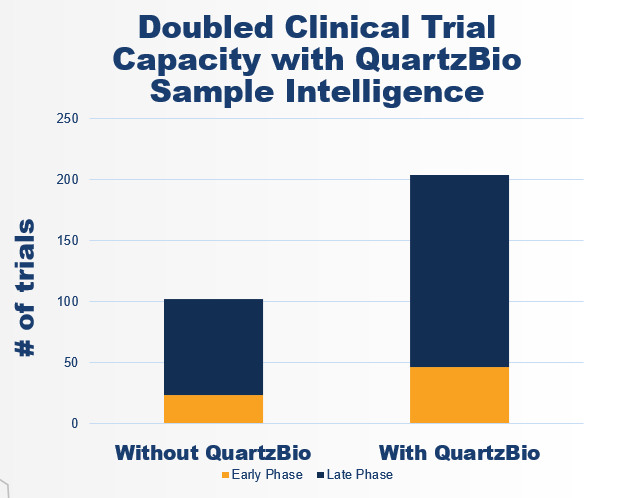
Figure 1. Increased capacity: By implementing QuartzBio, the client increased their study capacity by 100+ trials without adding headcount.
Financial Impact**
The efficiency gains translated directly into cost savings. Using a fully loaded FTE cost of $250,000 (as cited by the client) and the observed 85% reduction in sample data management effort, the savings per study ranged from $196K to $283K, with a reliable planning midpoint of approximately $239K per study.
Scaled across a portfolio of 100+ trials, that represents the potential for hundreds of millions of dollars in efficiency gains — without compromising quality or compliance.
Qualitative Outcomes
- Improved data quality, completeness, and timeliness of monitoring.
- Earlier detection of issues that previously went unnoticed, reducing the risk of patient data loss
- Strong cross-functional adoption, with 100% of users recommending the platform for continued use
In Their Words
At the close of the case study period, 100% of users reported that they would recommend the QuartzBio platform and Sample Intelligence solution. Here are a few of their comments:
“I was able to reduce manual reconciliation efforts and follow-up communications related to sample status and discrepancies.”
“Sample Intelligence (SI) was able to identify a pattern of data discrepancy– which identified a potential issue with our requisition form. That demonstrates the purpose of SI for our organization. No one had raised this as an issue previously–SI allowed us to identify this issue. This allowed us to fix a problem we wouldn’t have ever known about.”
Methodology
*Methodology used to calculate increased sample operations capacity:
Users reported the time required to complete key sample operations tasks before QuartzBio’s solution was implemented and again after implementation to quantify time savings. We then incorporated the proportion of a typical sample operations role dedicated to these tasks to determine realistic FTE impact. Finally, these reductions were applied across the existing trial portfolio to estimate the overall increase in capacity that the solution enabled.
**Methodology used to calculate financial impact:
We quantified the per-study financial impact of an 85% efficiency gain applied to sample data workflows. Using the typical team FTE range of 0.90 to 1.30 FTE, the reduction yields 0.765 to 1.105 FTE saved per study. When applying the fully loaded cost of $250,000 per FTE as cited by the client, the resulting savings ranged from $191,250 to $276,250 annually. The midpoint of this range—approximately $233,750—serves as a reliable single-value estimate for planning and forecasting.
Looking Ahead
This case study demonstrates that when sample data is unified, automated, and accessible in real time, teams can achieve 85% faster task completion, 100% missing sample detection, doubled study capacity, and more.
These savings emerge from reductions in manual reconciliations, vendor follow-up, data processing, sample inventory review, and cross-functional coordination. When scaled across multiple studies, this can create multi-million-dollar portfolio efficiencies, strengthen operational agility, reduce burnout risk, and enhance timelines for data-driven decisions.
Future case studies may explore additional downstream benefits, including reductions in manual errors, improved cross-functional collaboration, and faster overall R&D cycle times.
Learn how Sample Intelligence could amplify work for your team.
Reach out to our team for a 15-minute review of your current workflow at www.quartz.bio/contact-us.
Selected References
Protocol complexity and data volume scale up from Phase 1 → 3 (drives workload).
- Tufts CSDD has repeatedly documented the sharp rise in procedures, endpoints, and data points in Phases II–III; more data and procedures translate into higher operational burden on monitoring, data management, biospecimen logistics, and reconciliation. [https://www.clinicaltrialvanguard.com/conference-coverage/tufts-csdd-new-insights-on-the-clinical-trial-industry/]
- A 2025 TransCelerate + Tufts analysis across 105 Phase II/III protocols shows ~⅓ of procedures are non‑core—still work that sites/ops must manage—further inflating workload.
- Syntheses from ICON and CRIO echo Tufts’ numbers (more procedures, sites, endpoints; more amendments), linking rising complexity to greater operational effort.
Operational footprint expands with study size (sites, recruitment, duration).
- Nature’s analysis of 2,140 Phase III trials shows how large Phase III programs extend across more sites and recruit at slower per‑site rates—hallmarks of higher coordination and data/biomarker ops effort than earlier phases.
Roles that touch sample data are cross‑functional and scale with complexity.
- EMA’s GCP Inspectors’ Reflection Paper on labs analyzing clinical‑trial samples details required controls around labeling, receipt, storage, chain of custody, validation, and data/IT—work that expands with sample throughput and vendor networks typical of later phases. [ema.europa.eu]
- UKCRC guidance for Clinical Trial Units (CTUs) reinforces oversight and documentation expectations for labs and sample workflows under GCP—again indicating more moving parts = more effort.
Biospecimen management best practices formalize the non‑negotiable work (independent of ‘analytics’).
- NCI Best Practices for biospecimens and ISBER Best Practices for biorepositories enumerate the end‑to‑end activities—collection, processing, shipping, storage, tracking, QA/QC, data management—required to keep sample integrity and traceability; as counts/timepoints/vendors increase by phase, so do these activities.
Workload/acuity models from the field validate the need to budget FTE by complexity, not just headcount.
- ACRP’s peer‑reviewed article argues for complexity‑adjusted productivity models (beyond ‘patients per coordinator’)—the same rationale used to frame phase‑based FTE bands. [acrpnet.org]
- SWOG (NCI network) and Mayo Clinic posters show practical workload sizing and CRC effort tools—evidence that programs estimate effort as protocol/site/sample complexity rises.
Analysis of screening, treatment and follow-up expenses, biological treatment per-patient costs can exceed $100,000.
- Biology Insights – “Clinical Trial Cost per Patient: Key Drivers,” April 29, 2025.
- Average per‑patient costs across phases: – Phase I average: $136,783 per patient – Phase I/II average: $155,340 per patient Source: ProRelix Research – “Phase-by-Phase Clinical Trial Costs Guide,” May 5, 2025.
- Rare disease trial complexity and elevated operational costs: – Higher burdens due to limited patient pools, complex regimens, and recruitment challenges. Source: Rare Revolution Magazine – “Understanding the true cost of clinical trials,” August 4, 2025.