By Melinda Pautsch, Chief Commercial Officer, QuartzBio
I just returned from SCOPE 2026 with a familiar feeling—and a sharper sense of urgency. The conversations around biomarkers, biospecimens, and clinical operations weren’t about “doing things better” at the margins. They were about whether our operational foundations can keep up with what science—and regulators—are demanding.
Across sessions and panels, one idea kept resurfacing: biospecimens are no longer treated as physical materials moving through a supply chain. They’re treated as data—and that reframes everything from protocol design to chain of custody to the quality systems we need to prove we can trust what we generate.
Below are the themes that stood out, with pragmatic takeaways for biomarker operations, biospecimen operations, and clinical operations leaders who are trying to scale without taking on disproportionate risk.
1. “No more spreadsheets” isn’t a slogan—it’s a scalability requirement
If SCOPE had an award for “Most Vilified Operations Tool” it would be spreadsheets (followed closely by paper) as the silent accelerant of operational risk.
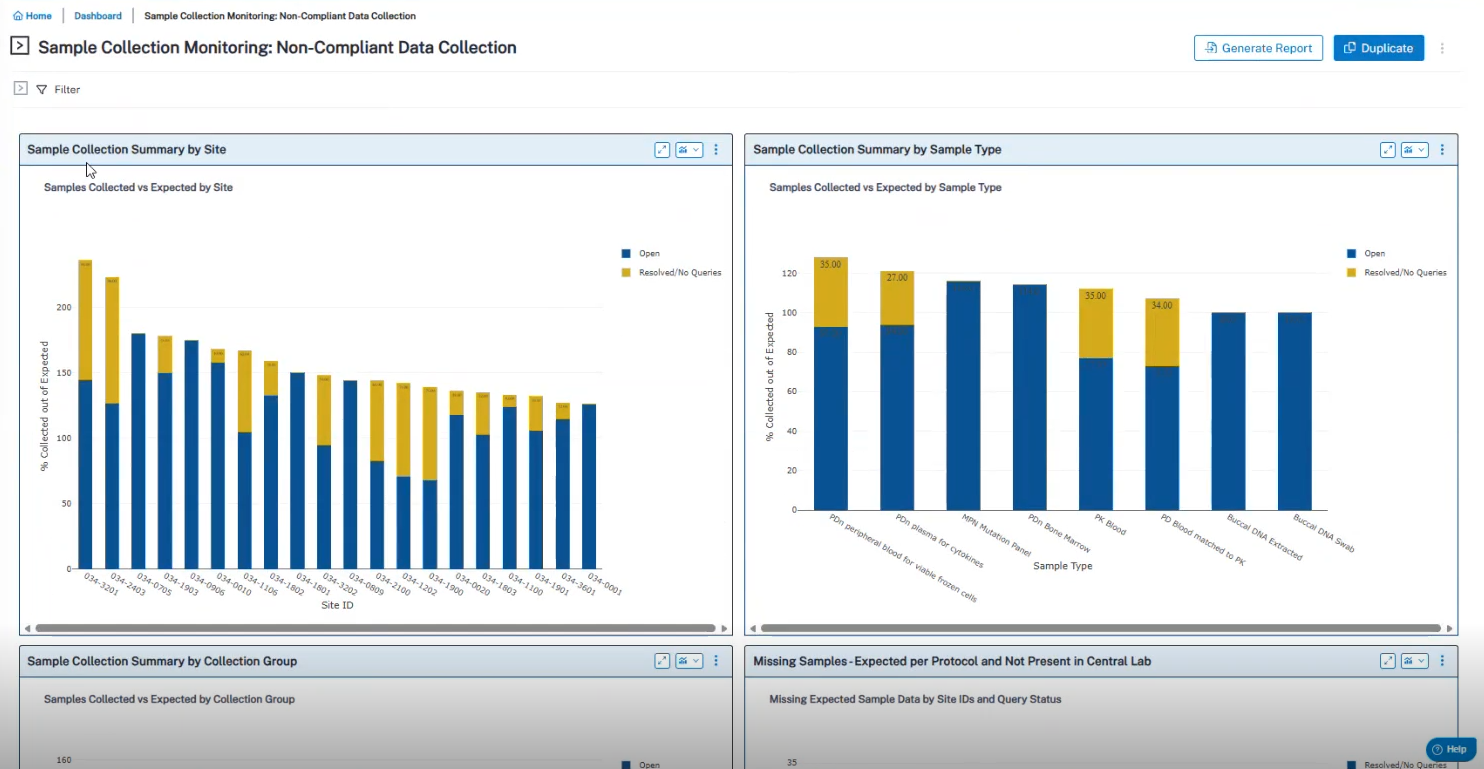
Brooke Samuelian of Syndax kicked off the topic of digitizing sample management, describing what many of us have lived: tracking specimens manually is time-consuming and error-prone, and failures cascade into missed collections, lost shipments, lab queries, protocol deviations, and even patient re-recruitment.
Michael Tanen of Merck was even more direct: moving away from Excel will be the single biggest leap forward—and with the rollout of the ICH E6(R3) GxP guideline, it won’t remain optional.
Operational takeaway: Digitization isn’t just about efficiency; it’s about avoiding preventable failure modes:
- Mislabeling, misrouting, or “orphaned” specimens
- Incomplete consent tracking that limits downstream use or creates compliance exposure
- Lack of real-time visibility that forces reactive escalation
- Manual updates that introduce inconsistency across stakeholders
Target outcomes: A digitized, connected sample workflow reduces the likelihood of wasted samples, protects patient intent, and improves first-time-right execution—the foundation for scalable biomarker strategy.
2. Risk management in modern clinical trials demands a proactive approach to operations
The compliance conversations at SCOPE, whether around E6(R3) or risk-based quality management in general, dove into the operational consequences of evolving regulations and increasing clinical trial complexity. Two points stood out.
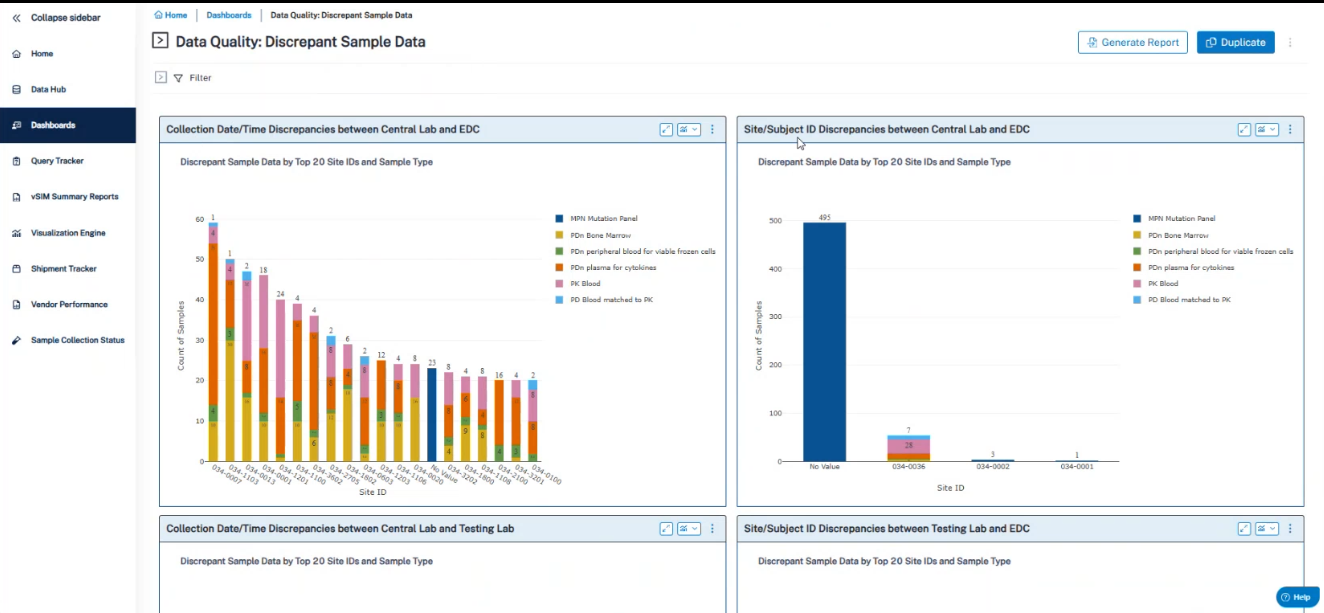
First: risk monitoring is no longer passive or reactive. E6(R3) emphasizes active risk monitoring and expects sponsors and labs to show linkages and chain of custody.
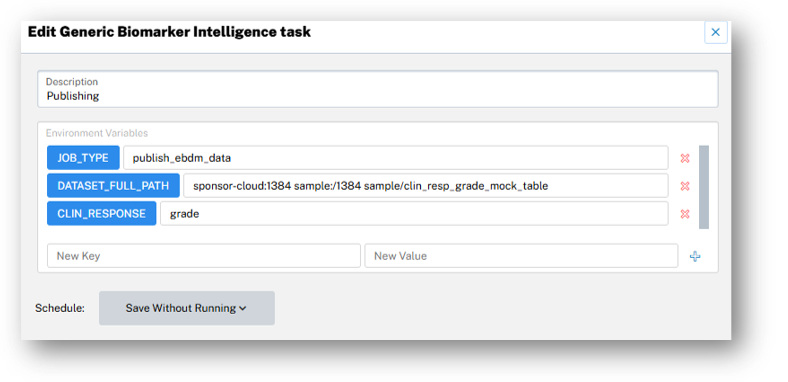
At his presentation on Day 2 of SCOPE, QuartzBio’s CTO and Co-Founder, Tobi Guennel, PhD, showed how an agentic framework, powered by proactive and prescriptive AI, transforms clinical operations.
These AI agents proactively track sample logistics, flag inconsistencies, and issue alerts with actionable, corrective steps, reducing issues by >80%, accelerating milestones, and saving on average $150,000 per trial. (Note — the metrics presented were appreciated by the audience in an industry where the ROI of agentic frameworks is still elusive to many.)
Second: Speakers agreed that building a compliant foundation is crucial. If maintaining end-to-end visibility across the sample chain of custody isn’t addressed during protocol design, teams are likely to inherit the downstream consequences—with fewer options and higher cost.
Operational takeaway: Treat biospecimen strategy as a front-end design input, not an execution detail:
- Identify which samples are “critical” vs. nice-to-have
- Build chain-of-custody expectations into vendor and lab selection
- Make reconciliation a mandatory step for maintaining data quality
Target outcomes: Traceability and governance reduce inspection risk, protect data integrity, and prevent downstream surprises that derail timelines.
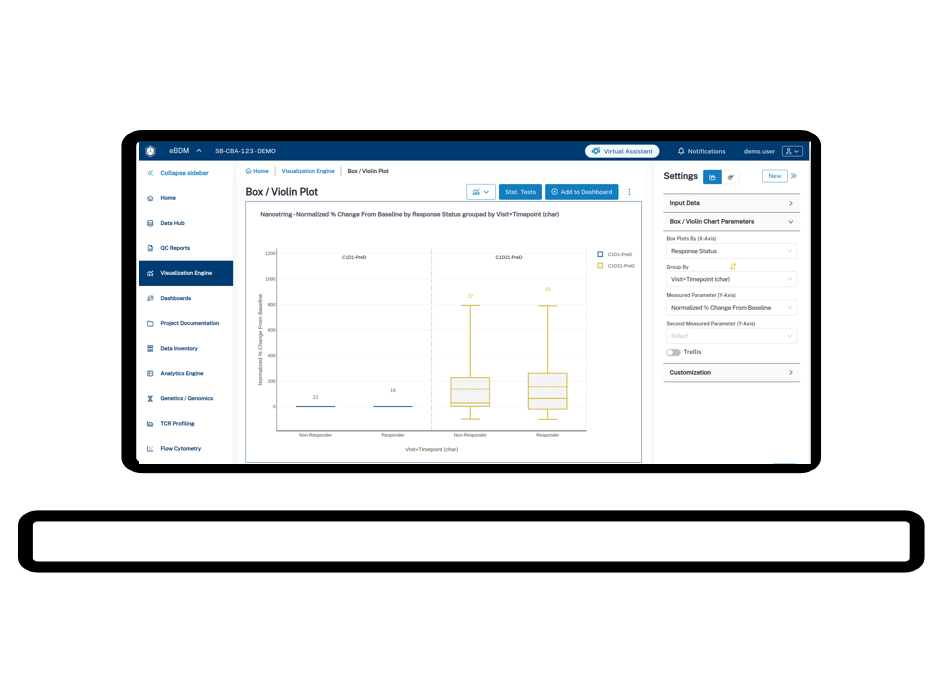
3. AI doesn’t fix broken, disconnected data
Several AI-focused discussions were refreshingly grounded. One panelist captured what many are feeling: before we can use AI to unlock visibility, analytics, and actionable insights, we need to build interoperable ecosystems of clean, connected operational data. AI doesn’t fix broken data. It amplifies whatever foundation you have.
Operational takeaway: Prioritize technologies that FAIRify data at its foundation over pilots that promise insights without addressing underlying data chaos.
- Avoid “death by pilots” that never scale or win sustained budget
- Connect the data value chain: consistent metadata, governed transfers, and linked workflows
- Make it easy for teams to find information, validate status, and forecast availability
Target outcomes: Deep data FAIRification improves on-time delivery, reduces fire drills, and enables smarter planning—so teams can redirect effort from tracking work to advancing science.
Sample Intelligence: the operating system for biomarker and biospecimen strategy
I’ll keep one quote from SCOPE as my guiding light for 2026:
“Every sample is more than a vial—it carries a patient’s hope, a researcher’s trust, and the promise of better medicine. Tracking it with precision is not just compliance; it’s compassion.” –quoted by Dmitri Mikhailov, PhD, Novartis Institutes for BioMedical Research.
Operational leaders are being asked to deliver that precision at scale—across protocols that are more complex, ecosystems that are more distributed, and regulatory expectations that are more explicit. The path forward isn’t a single tool or another point solution. It’s connected Sample Intelligence: end-to-end visibility, governed chain of custody, standardized metadata, and interoperable systems that treat biospecimens as the strategic data assets they are.
Let’s talk about what “future-ready” looks like for your team
If you’re evaluating how ready your current operations are for E6(R3), advanced analytics, or simply the next wave of complexity, I’d welcome a conversation. Reach out to connect with me about:
- Evaluating your current sample management workflows and where friction is hiding risk
- Assessing gaps across your biomarker and clinical data ecosystem (visibility, governance, reconciliation)
- Exploring how greater sample intelligence can support scalable, inspection-ready, decision-confident operations
Email quartzbio-sales@precisionformedicine.com or visit www.quartz.bio/contact.