Video: Centralized, harmonized data overcomes silos, revealing insights.
Overcome data silos
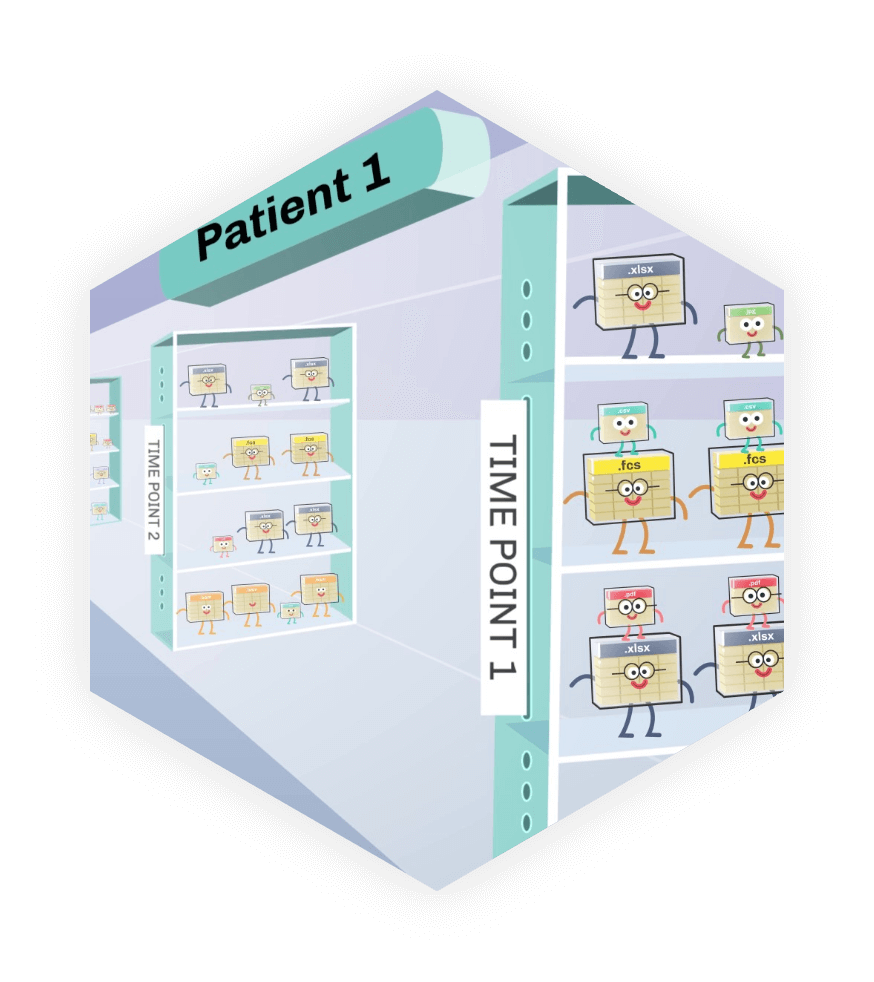
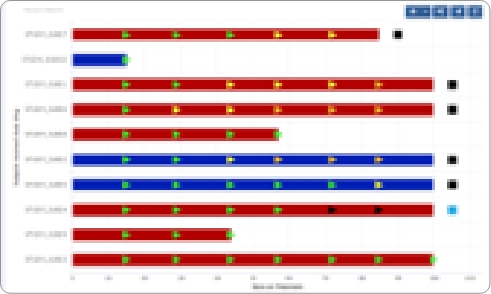
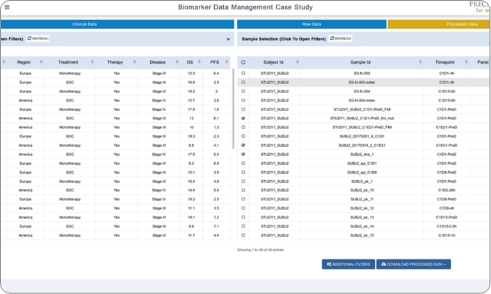
QuartzBio’s enterprise Biomarker Data Management solution links assay and clinical data across file types from any assay technology–transcriptomics, genomics, proteomics, flow cytometry, imaging, and more. Users across multiple teams can explore, collaborate on, and report their data using our suite of analytics and reporting solutions.
Here’s how different teams are using the solution:
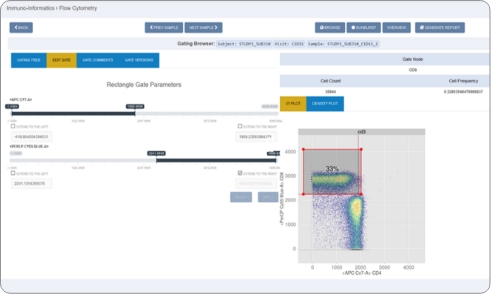
- Biomarker Operations: Systematically verify data quality, consistency, and availability with automated checks
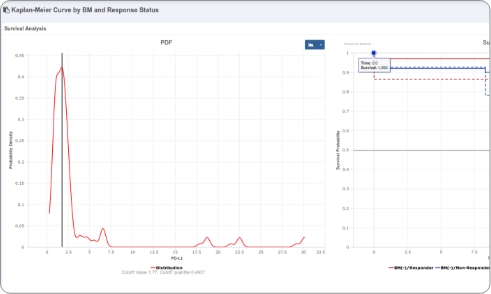
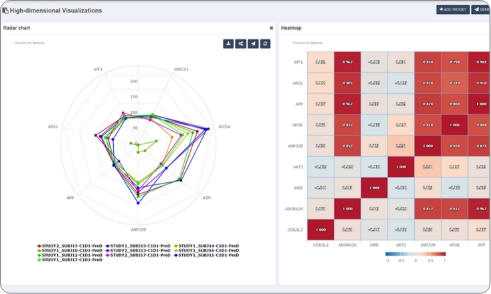
- Translational Researchers: Explore and visualize biomarker trends across cohorts, subjects, and timepoints and perform exploratory analyses to surface signals of interest
- Bioinformatics Teams: Generate visualizations and connect data to existing tools (e.g., Spotfire, Prism)